- Record: found
- Abstract: found
- Article: found
Identification of Two Novel Anti-Fibrotic Benzopyran Compounds Produced by Engineered Strains Derived from Streptomyces xiamenensis M1-94P that Originated from Deep-Sea Sediments
Read this article at
Abstract
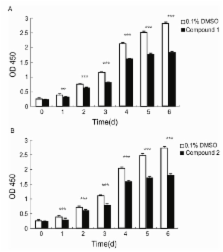
The benzopyran compound obtained by cultivating a mangrove-derived strain, Streptomyces xiamenensis strain 318, shows multiple biological effects, including anti-fibrotic and anti-hypertrophic scar properties. To increase the diversity in the structures of the available benzopyrans, by means of biosynthesis, the strain was screened for spontaneous rifampicin resistance (Rif), and a mutated rpsL gene to confer streptomycin resistance (Str), was introduced into the S. xiamenensis strain M1-94P that originated from deep-sea sediments. Two new benzopyran derivatives, named xiamenmycin C ( 1) and D ( 2), were isolated from the crude extracts of a selected Str-Rif double mutant (M6) of M1-94P. The structures of 1 and 2 were identified by analyzing extensive spectroscopic data. Compounds 1 and 2 both inhibit the proliferation of human lung fibroblasts (WI26), and 1 exhibits better anti-fibrotic activity than xiamenmycin. Our study presents the novel bioactive compounds isolated from S. xiamenensis mutant strain M6 constructed by ribosome engineering, which could be a useful approach in the discovery of new anti-fibrotic compounds.
Related collections
Most cited references24
- Record: found
- Abstract: found
- Article: not found
Plant-derived leading compounds for chemotherapy of human immunodeficiency virus (HIV) infection.
- Record: found
- Abstract: found
- Article: not found
Induction of actinorhodin production by rpsL (encoding ribosomal protein S12) mutations that confer streptomycin resistance in Streptomyces lividans and Streptomyces coelicolor A3(2).

- Record: found
- Abstract: found
- Article: found