- Record: found
- Abstract: found
- Article: found
Metformin modulates hyperglycaemia-induced endothelial senescence and apoptosis through SIRT1
Read this article at
Abstract
Background and Purpose
Endothelial dysfunction can be detected at an early stage in the development of diabetes-related microvascular disease and is associated with accelerated endothelial senescence and ageing. Hyperglycaemia-induced oxidative stress is a major contributing factor to the development of endothelial dysfunction. Clinical data indicate that the hypoglycaemic agent, metformin, has an endothelial protective action; however, its molecular and cellular mechanisms remain elusive. In the present study, we have investigated the protective effect of metformin during hyperglycaemia-induced senescence in mouse microvascular endothelial cells (MMECs).
Experimental Approach
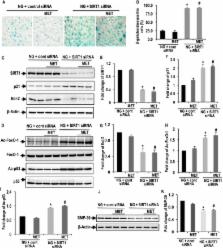
MMECs were cultured in normal glucose (11 mM) and high glucose (HG; 40 mM) in the presence and absence of metformin (50 μM) for 72 h. The expression of sirtuin-1 (SIRT1) and senescence/apoptosis-associated markers was determined by immunoblotting and immunocyto techniques. SIRT1 expression was inhibited with appropriate siRNA.
Key Results
Exposure of MMECs to HG significantly reduced SIRT1 protein expression, increased forkhead box O1 (FoxO-1) and p53 acetylation, increased p21 and decreased Bcl2 expression. In addition, senescence-associated β-galactosidase activity in MMECs was increased in HG. Treatment with metformin attenuated the HG-induced reduction of SIRT1 expression, modulated the SIRT1 downstream targets FoxO-1 and p53/p21, and protected endothelial cells from HG-induced premature senescence. However, following gene knockdown of SIRT1 the effects of metformin were lost.
Related collections
Most cited references41
- Record: found
- Abstract: found
- Article: not found
Acetylation is indispensable for p53 activation.
- Record: found
- Abstract: found
- Article: not found
Deacetylation of FoxO by Sirt1 Plays an Essential Role in Mediating Starvation-Induced Autophagy in Cardiac Myocytes.
- Record: found
- Abstract: found
- Article: not found