- Record: found
- Abstract: found
- Article: found
Advanced Glycation End Products Enhance Macrophages Polarization into M1 Phenotype through Activating RAGE/NF- κB Pathway
Read this article at
Abstract
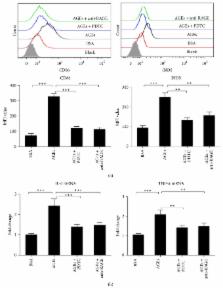
Atherosclerotic lesions are accelerated in patients with diabetes. M1 (classically activated in contrast to M2 alternatively activated) macrophages play key roles in the progression of atherosclerosis. Since advanced glycation end products (AGEs) are major pathogenic factors and active inflammation inducers in diabetes mellitus, this study assessed the effects of AGEs on macrophage polarization. The present study showed that AGEs significantly promoted macrophages to express IL-6 and TNF- α. M1 macrophage markers such as iNOS and surface markers including CD11c and CD86 were significantly upregulated while M2 macrophage markers such as Arg1 and CD206 remained unchanged after AGEs stimulation. AGEs significantly increased RAGE expression in macrophages and activated NF- κB pathway, and the aforementioned effects were partly abolished by administration of anti-RAGE antibody or NF- κB inhibitor PDTC. In conclusion, our results suggest that AGEs enhance macrophage differentiation into proinflammatory M1 phenotype at least partly via RAGE/NF- κB pathway activation.
Related collections
Most cited references33
- Record: found
- Abstract: found
- Article: not found
Role of advanced glycation end products in cellular signaling☆
- Record: found
- Abstract: found
- Article: not found