- Record: found
- Abstract: found
- Article: found
Evidence-based clinical practice guidelines for inflammatory bowel disease 2020

Read this article at
Abstract
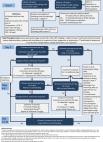
Inflammatory bowel disease (IBD) is a general term for chronic or remitting/relapsing inflammatory diseases of the intestinal tract and generally refers to ulcerative colitis (UC) and Crohn’s disease (CD). Since 1950, the number of patients with IBD in Japan has been increasing. The etiology of IBD remains unclear; however, recent research data indicate that the pathophysiology of IBD involves abnormalities in disease susceptibility genes, environmental factors and intestinal bacteria. The elucidation of the mechanism of IBD has facilitated therapeutic development. UC and CD display heterogeneity in inflammatory and symptomatic burden between patients and within individuals over time. Optimal management depends on the understanding and tailoring of evidence-based interventions by physicians. In 2020, seventeen IBD experts of the Japanese Society of Gastroenterology revised the previous guidelines for IBD management published in 2016. This English version was produced and modified based on the existing updated guidelines in Japanese. The Clinical Questions (CQs) of the previous guidelines were completely revised and categorized as follows: Background Questions (BQs), CQs, and Future Research Questions (FRQs). The guideline was composed of a total of 69 questions: 39 BQs, 15 CQs, and 15 FRQs. The overall quality of the evidence for each CQ was determined by assessing it with reference to the Grading of Recommendations Assessment, Development and Evaluation approach, and the strength of the recommendation was determined by the Delphi consensus process. Comprehensive up-to-date guidance for on-site physicians is provided regarding indications for proceeding with the diagnosis and treatment.
Related collections
Most cited references398
- Record: found
- Abstract: found
- Article: not found
Infliximab for induction and maintenance therapy for ulcerative colitis.
- Record: found
- Abstract: found
- Article: found
British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults
- Record: found
- Abstract: found
- Article: not found