- Record: found
- Abstract: found
- Article: found
Editorial: Home Cage-Based Phenotyping in Rodents: Innovation, Standardization, Reproducibility and Translational Improvement
editorial
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Novel and emerging technologies, such as home cage monitoring (HCM) systems, permit
24/7 collection of behavioral data under undisturbed conditions. HCM minimizes the
impact of stressors that arises from human interaction or the testing in novel test
environments, which may bias readout parameters (science) and/or affect animal welfare
since science and welfare are highly interdependent. There is increasing evidence
that laboratory animals living under enriched and group-housed conditions display
a behavioral repertoire that is much richer as compared to what is observed in classical
behavioral experiments (e.g., open field, total distance traveled and number of center/corner
visits). Behavioral phenotyping experiments performed by classical tests show limited
replicability due to idiosyncratic results to a particular laboratory (Crabbe et al.,
1999). With this editorial about our Research Topic we try to highlight advances in
technologies in HCM that are relevant for animal welfare, scientific aspects or both,
such as the development of novel biomarkers (see Baran et al.) that can lead to better
translational approaches in different animal welfare and scientific areas as described
below.
Definition of HCM Systems
HCM occurs in a cage where the animal spends more than a limited period of time (≥24
h) to either perform certain behavioral tasks or display physiological responses.
However, proper HCM should be differentiated from benchtop technologies as highlighted
by Baran et al. The need for a consensus across the neuroscience community recently
led to the initiation of two work groups mainly in North America (https://www.na3rsc.org/tdb/a)
and Europe (https://www.cost.eu/actions/CA20135/) that now try to harmonize the definitions
of HCM, provide examples, generate guidelines and try to identify standards for improved
comparability of results and new developments.
Reproducibility and Methods in Behavioral Science
The reproducibility crisis in animal research is a well-known issue (Richter and von
Kortzfleisch, 2020). Can automatization increase reproducibility of animal research?
Richter suggests that controlled heterogenization in combination with HCM may lead
to increase constructive and predictive validity. Along the line of data reproducibility,
Engelbeen et al. tried to replicate findings in the mdx mouse model of Duchenne muscular
dystrophy by testing mice in a standard battery of behavioral tests using both conventional
and HCM methods. In contrast to previous findings using only female mdx mice, a learning
deficit could not be reproduced suggesting an interaction of genetic and environmental
factors contributing to cognitive performance differences in mdx mice.
According to Voikar and Gaburro and Baran et al., a combination of different technologies
can help identify biomarkers that have the potential for transfer to the clinic. Including
data that cover 99% of an animal's time and obtaining such data from the more complex
situation of group-housing may increase the power of such approaches. New challenges
range from 24/7 data management, data analysis tools (software tools), interpretation
of large, unstructured data sets up to IT/cybersecurity issues. This means that not
only scientists are necessary in a successful implementation of HCM systems.
Behavioral video monitoring has a long history as shown by Baran et al. and in the
review by Grieco et al. These articles provide an overview of the use of video monitoring,
the interpretation of data generated, and the scope that can be obtained from an expanded
use of video monitoring technology. More recent developments advancing this technique
are explained by Gharagozloo et al. who show that complex and more naturalistic behaviors
can be studied through machine learning approaches and artificial intelligence.
Alternatives to video monitoring with specific applications to learning and memory
are described by Voikar and Gaburro, other technologies are applied in substance use
disorders as described by Iman et al. Specifically, RFID-based technology to recognize
event-based animal behavior can be employed. Such technology allows interpretations
in the context of reward learning and more complex behaviors such as sociability.
The use of RFID transponders in group-housed mice also allows to study lifetime changes
in animal behaviors in normal and pathological conditions which may address important
scientific questions such as the interaction between cognition and aging Kahnau et
al. which is fundamental especially in neurodegenerative disease progression.
Animal Welfare
Four articles address the connection between animal welfare and behavioral experimental
outcome. This includes using behavioral outcome to classify the severity level of
a treatment in the context of animal welfare. In fact, simple routine operation such
as cage-change can increase physiological parameters (heart rate, locomotion) for
90 min up to a few hours (Stiedl et al., 2004; Pernold et al., 2019). Analyzing the
physiological response, specifically heart rate, to evaluate the impact of surgery
with radio-telemetry for a neuroscience rat model, was important to assess when the
animals could be exposed to a battery of behavioral tests as reported by Wassermann
et al. A follow-up study from the same group by Zentrich et al. used spontaneous activity
as marker of animal welfare. Here, an acute experimental colitis mouse model, starting
from the highest level of severity as established by a clinical score (10 different
parameters), expressed a reduction in locomotion that remained significantly suppressed
until the mice recovered from the treatment indicating that HCM can be used for routine
severity assessment in biomedical research.
The effect of ambient illumination on the behavior of animals has not been systematically
studied in the animal facility where animals are bred and kept. Yet, it is well known
that light conditions can dramatically affect the expression of various behaviors
in visual behavior tests. Therefore, Steel et al. systematically evaluated the spontaneous
activity depending on the position of the cage in the rack. Cage position relative
to the position of the room lights had a dramatic effect on 24/7 activity in the HCM
as measured by different parameters. Single parameters differed up to 15-fold between
cages in top (higher light intensity) vs. bottom rack position (lower light intensity).
For cage position from left to right there was still a 3-fold difference in light
intensity. This suggests that for increased uniformity and reproducibility of cage
rack studies, each cage should have its own controllable light source. Interestingly,
animals in red tinted cages, which block most visible light, did not display such
cage variation in activity between cage locations. However, they showed a reduced
diurnal activity level as compared to animals in standard transparent cages. This
shows trade-offs between positive aspects for animals vs. care takers and visual daily
checks.
Moore and Brook used HCM-derived activity to assess the effect of replacing an individual
companion female by a different one on spontaneous locomotion of males in their cage.
Using such technology revealed that replacing a female resulted in an increase in
activity for up to 4 h. This was followed by a compensatory reduction in activity
during the subsequent dark phase as potential sign of fatigue. A resulting refinement
suggestion is that cage mate replacements should occur toward the end of the light-phase
when animal activity increases, probably impacting less on the normal circadian activity.
Neurodegenerative Models
Despite differences in their clinical manifestations, neurodegenerative diseases such
as Alzheimer, Parkinson and others share common symptoms both at the clinical and
at the preclinical level. An example are alterations in resting bouts in a model of
Amyotrophic lateral sclerosis (ALS). Here, resting bouts were used as an index of
reduced sleep to establish a novel biomarker called rest disruption index (RDI). The
RDI indicates the reduction of sleep in SOD1G93A mice at 16–18 weeks of age. This
finding correlates with symptoms in typical behavioral tests such as the grid test
as described by Golini et al.
Another approach based on a HCM with attached automated figure-8-maze (F8M) was used
to test transgenic APPswe/PSEN1dE9 (APP/PS1) mice in the delayed alternation task
to gain water as reward in a longitudinal manner from 2–6 months of age by van Heusden
et al. Only 6-month-old transgenic APP/PS1 mice displayed an increase in the number
of consecutive incorrect responses. The F8M approach indicates the possibility to
monitor performance repeatedly over months. However, the expected cognitive decline
was not detected as early as expected, since an earlier study (Vegh et al., 2014)
reported the onset of cognitive impairment in the water maze task at 4 months of age
in APP/PS1 mice. Particularly in animal models with increased anxiety-like phenotype
such as APP/PS1 mice, deficits in tasks such as the water maze (Wolfer et al., 1998)
may be the result of confounding effects of emotionally challenging experimental conditions
on cognition in complex tasks (Diamond et al., 2007). Thus, HCM-based approaches are
better suited to minimize unspecific effects of emotionally challenging conditions
on cognition as demonstrated by another elegant HCM approach in 3-month-old APP/PS1
mice with higher sensitivity of visual discrimination to prefrontal cortex dysfunction
than water maze performance (van den Broeck et al., 2021).
Learning and Memory: Visual, Olfactory and Auditory Cues
Visual and olfactory cues are typically very difficult to be analyzed within HMC systems.
In one of the very first articles of our Research Topic Wooden et al. the researchers
substantially improved the object recognition test by applying an inexpensive 3-step
approach that allows to adjust the experimental conditions according to the model
for providing more robust results.
Advanced testing procedures in the learning, memory, and cognitive domain are based
on single animal performance in an operant chamber. When an operant chamber is connected
to the home cage, a form of behavioral enrichment is achieved that simultaneously
allows 24/7 voluntary testing of RFID-tagged mice. This approach of Caglayan et al.
used olfactory stimuli to investigate learning set acquisition. Full automation in
this context eliminated human interaction and permitted individualized training schedules
where a specific mouse could enter to the next level of training based on individual
performance. This successfully allowed investigating higher-order cognitive function
in the home cage.
In their second study, Caglayan et al. used a home cage connected with a gating mechanism
to the operant chamber to perform the stop-signal-task for assessing symptoms of attention
deficit disorders and schizophrenia. The automated HCM approach required only minimal
experimenter involvement, reduced training time for the mice by about 20%, and importantly,
considerably improved task sensitivity.
Fear and Anxiety
Fear and anxiety are commonly studied with short experiments that cannot be applied
repeatedly, and therefore, do not allow the analysis of disease progression or the
assessment of long-term treatment. Schuessler et al. customized an experimental chamber
consisting of a safe nest and a foraging area with an operant lever, food port, water
spout and shock grid floor. Foot shock was delivered specifically in the area where
food and water were available. Using this “Risky Closed Economy” approach, fear and
anxiety-related behavior were studied nearly 24/7 for extended periods as described,
indicating flexibility based on decision-making when and how much to forage. This
is a more naturalistic foraging scenario under controlled threat condition. Similarly,
RFID-chipped transgenic mice in a partially automated HCM can be studied for their
social, cognitive behavior in a complex environment that helps researchers identify
the gene x environment interactions for affective and cognitive consequences of psychiatric
disease as shown by Volkmann et al..
Stroke
Most stroke models in rodents are induced via transient occlusion of the middle cerebral
artery and are behaviorally characterized through standard motor tests. However, repetition
of such tests might lead to habituation and changes in performance. Therefore, researchers
tried to identify hallmarks after stroke induction on locomotor activity in HCM. A
new open-source tracking software allowed calculating distance traveled, speed and
also turning behavior since stroke models have the tendency to move more unilaterally
if untreated. Here, recovery from stroke was not augmented by dietary induction as
shown previously in humans by Shenk et al. A follow-up paper from the same group determined
whether voluntary wheel running in the HCM would induce a faster recovery from stroke.
In fact, stroke-induced mice with running wheel had a better recovery from stroke
in terms of motor skills as well as increased functional connectivity, cerebral blood
flow, and vascular quality as reported by Lohkamp et al..
Viral Infection Symptoms
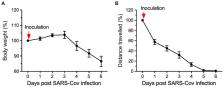
HCM also served in the evaluation of mice exposed to SARS-CoV-2. K18-hACE2 transgenic
mice, harboring the humanized angiotensin-converting enzyme 2 (ACE2) receptor to which
the spike protein binds, were used to assess the pathophysiology induced by SARS-Cov-2.
Typically, in infectious disease studies using mice both viral titer and body weight
are monitored. However, SARS-Cov-2 infections in humans cause fatigue rather than
body weight loss. Using a HCM system, K18-hACE2 transgenic mice infected with SARS-CoV-2
were evaluated daily for body weight as well as locomotor activity (see Figure 1).
These results demonstrate the potential of how automated HCM can aid both, animal
welfare assessment and viral infection-induced symptom detection of disease in mice
similar as in humans (Gaburro, 2021; Kaufmann et al., 2022).
Figure 1
Eight single-housed K18-hACE2 (C57Bl/6J background) mice were exposed to the SARS-CoV-2
virus (2.8 × 104 TCID50/ml). Changes in body weight (A) and total distance traveled
(B) were assessed for six days relative to pre-inoculation values (100%). After viral
exposure (inoculation), body weight dropped significantly on day 4, whereas the distance
traveled was already significantly reduced by 30% on day 1 and reduced by almost 90%
on day 4 indicating substantially higher sensitivity of locomotor activity than body
weight change (Gaburro, 2021; modified from Kaufmann et al., 2022).
Energy Expenditure and Circadian Locomotor Activity
A survey of 30 mouse strains by König et al. indicated significant effects of strain,
gender and circadian phase on voluntary physical activity and energy expenditure in
automated HCM. This study highlights that naturally occurring genetic variation modulates
various innate activity behaviors, food intake and energy expenditure in mouse strains.
From HCM Based-Exploration of Mice Back to Human Diagnostic
An intriguing development originates from phenotyping studies of predominantly mouse
behavior based on the long-standing collaboration between Ilan Golani and Yoav Benjamini
(e.g., Fonio et al., 2009) consisting of computational exploratory data analysis methodology
including videotaping, tracking, and customized data analysis. This has now been applied
to human pre-walking infants in the interdisciplinary approach by Frostig et al. aimed
to examine the organization of infant exploration in a novel setting. Here, the stationary
mother serves as “home-base” reference point to characterize the exploration patterns
of typically developing infants, but does not serve, or serves much less as a reference,
for non-typically developing infants. This approach identified profound differences
between typically and non-typically developing human infants with respect to their
excursions from the mother as important reference point. Thus, this approach may serve
as novel assay to screen for deviating child development with very early diagnostic
potential at 8–18 months, as observed in autism spectrum disorder, if replicated and
extended on a larger scale.
Conclusion
The interest toward the Research Topic on HCM and the still widely diverging avenues
of development suggest that this field is just at the beginning of expansion and far
from maturation. This general approach could set the basis for novel studies in which
digital libraries can be constructed for control animals which eventually could contribute
to a reduction of animal use in research. HCM approaches will provide for a better
characterization of normal vs. deviant behavior including better “symptom progression”
and earlier recognition of potential diagnostic features for improved animal welfare
and science.
Author Contributions
SG, OS, YW, and JK wrote the manuscript and revised it. ML revised the manuscript.
All authors contributed to the article and approved the submitted version.
Funding
This work was supported by a NIH grant MH099073 (JK).
Conflict of Interest
SG works as scientific director at Tecniplast S.p.A. ML works as Chief Executive Officer
at Sylics BV. The remaining authors declare that the research was conducted in the
absence of any commercial or financial relationships that could be construed as a
potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily
represent those of their affiliated organizations, or those of the publisher, the
editors and the reviewers. Any product that may be evaluated in this article, or claim
that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Related collections
Most cited references11
- Record: found
- Abstract: found
- Article: not found
Genetics of mouse behavior: interactions with laboratory environment.
J Crabbe, D Wahlsten, B C Dudek (1999)

- Record: found
- Abstract: found
- Article: found
Reducing hippocampal extracellular matrix reverses early memory deficits in a mouse model of Alzheimer’s disease
Marlene Végh, Céline M Heldring, Willem Kamphuis … (2014)