- Record: found
- Abstract: found
- Article: found
Independently paced Ca 2+ oscillations in progenitor and differentiated cells in an ex vivo epithelial organ
Read this article at
ABSTRACT
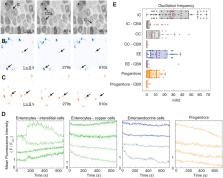
Cytosolic Ca 2+ is a highly dynamic, tightly regulated and broadly conserved cellular signal. Ca 2+ dynamics have been studied widely in cellular monocultures, yet organs in vivo comprise heterogeneous populations of stem and differentiated cells. Here, we examine Ca 2+ dynamics in the adult Drosophila intestine, a self-renewing epithelial organ in which stem cells continuously produce daughters that differentiate into either enteroendocrine cells or enterocytes. Live imaging of whole organs ex vivo reveals that stem-cell daughters adopt strikingly distinct patterns of Ca 2+ oscillations after differentiation: enteroendocrine cells exhibit single-cell Ca 2+ oscillations, whereas enterocytes exhibit rhythmic, long-range Ca 2+ waves. These multicellular waves do not propagate through immature progenitors (stem cells and enteroblasts), of which the oscillation frequency is approximately half that of enteroendocrine cells. Organ-scale inhibition of gap junctions eliminates Ca 2+ oscillations in all cell types – even, intriguingly, in progenitor and enteroendocrine cells that are surrounded only by enterocytes. Our findings establish that cells adopt fate-specific modes of Ca 2+ dynamics as they terminally differentiate and reveal that the oscillatory dynamics of different cell types in a single, coherent epithelium are paced independently.
Abstract
Summary: Live imaging of the Drosophila intestinal epithelium reveals that stem cells and their terminal progeny exhibit distinct, fate-specific Ca 2+ oscillations that are paced independently of each other.
Related collections
Most cited references56
- Record: found
- Abstract: found
- Article: not found
Fiji: an open-source platform for biological-image analysis.
- Record: found
- Abstract: found
- Article: not found