- Record: found
- Abstract: found
- Article: found
Transgenic viral expression of PH-20, IL-12, and sPD1-Fc enhances immune cell infiltration and anti-tumor efficacy of an oncolytic virus

Read this article at
Abstract
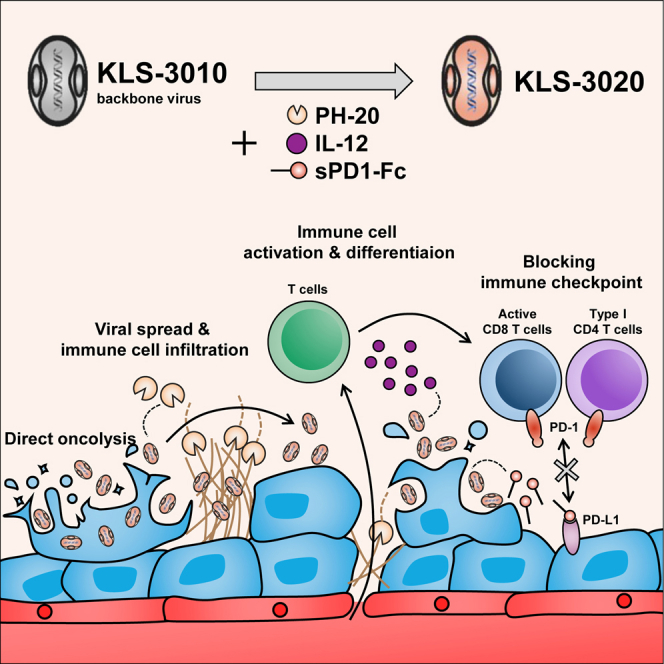
Oncolytic viruses are of significant clinical interest due to their ability to directly infect and kill tumors and enhance the anti-tumor immune response. Previously, we developed KLS-3010, a novel oncolytic virus derived from the International Health Department-White (IHD-W) strain vaccinia virus, which has robust tumoricidal effects. In the present study, we generated a recombinant oncolytic virus, KLS-3020, by inserting three transgenes (hyaluronidase [PH-20], interleukin-12 [IL-12], and soluble programmed cell death 1 fused to the Fc domain [sPD1-Fc]) into KLS-3010 and investigated its anti-tumor efficacy and ability to induce anti-tumor immune responses in CT26.WT and B16F10 mouse tumor models. A single injection of KLS-3020 significantly decreased tumor growth. The roles of the transgenes were investigated using viruses expressing each single transgene alone and KLS-3020. PH-20 promoted virus spread and tumor immune cell infiltration, IL-12 activated and reprogrammed T cells to inflammatory phenotypes, and sPD1-Fc increased intra-tumoral populations of activated T cells. The tumor-specific systemic immune response and the abscopal tumor control elicited by KLS-3020 were demonstrated in the CT26.WT tumor model. The insertion of transgenes into KLS-3020 increased its anti-tumor efficacy, supporting further clinical investigation of KLS-3020 as a novel oncolytic immunotherapy.
Graphical abstract
Abstract
Kim and colleagues improved their previous recombinant oncolytic vaccinia virus by arming it with PH-20, IL-12, and soluble PD1-Fc, leading to the significantly further increase of the number of cytotoxic T cells activated in tumor and marked suppression of tumor growth against solid tumors such as colon cancer and melanoma.
Related collections
Most cited references66

- Record: found
- Abstract: found
- Article: found
GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses
- Record: found
- Abstract: found
- Article: not found
Improved Survival with Ipilimumab in Patients with Metastatic Melanoma
- Record: found
- Abstract: found
- Article: not found