- Record: found
- Abstract: found
- Article: found
Neural and central mechanisms of kidney fibrosis after relief of ureteral obstruction
Read this article at
Summary
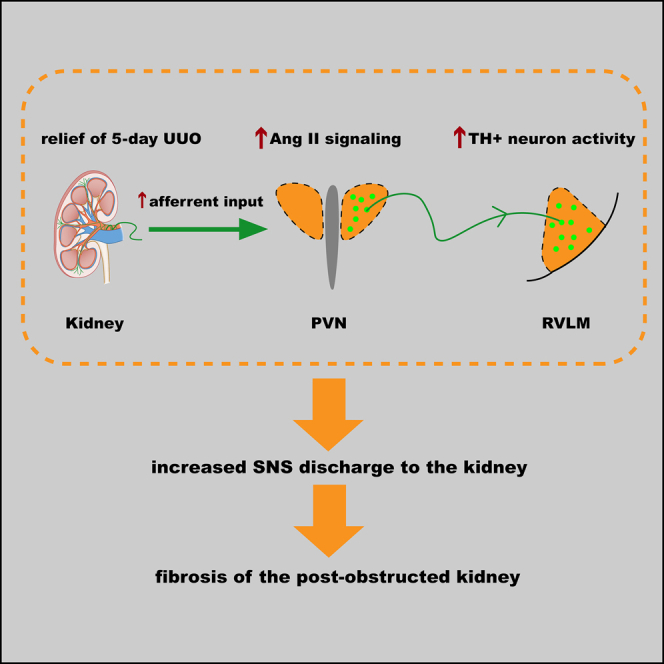
Obstructive uropathy from nephrolithiasis remains a leading cause of end-stage kidney disease. Mechanisms of kidney fibrosis after relief of ureteral obstruction represent opportunities for therapeutic intervention. Here, in mouse models of ureteral obstruction, we have combined methods of virus tracing and optogenetic techniques to identify an overactive central pathway in the paraventricular nucleus (PVN)-rostral ventrolateral medulla (RVLM) that determines the fibrotic fate of kidney after relief of the obstruction. The overactive pathway is driven by kidney afferent nerves that activate angiotensin II signaling in RVLM-projecting PVN neurons to drive sympathetic discharge back to the kidney. This causes the kidney to undergo fibrosis with loss of function. Blockade of sympathetic traffic or deletion of AT1a in PVN preserves the structure of the post-obstructed kidney. Human post-obstructed kidneys also demonstrate evidence of increased sympathetic nerve activity associated with a fibrotic outcome. Manipulating these neural elements is a potential treatment strategy.
Graphical abstract
Highlights
-
•
An overactive central pathway in PVN-RVLM mediates kidney fibrosis after UUO relief
-
•
Kidney afferent inflow activates Ang II signaling in RVLM-projecting PVN neurons
-
•
Activated PVN-RVLM pathway enhances SNS discharge to the kidney to promote fibrosis
-
•
Blocking SNS or deleting PVN AT1a preserves structure of post-obstructed kidneys
Abstract
Biological sciences; Neuroscience; Systems neuroscience
Related collections
Most cited references44
- Record: found
- Abstract: found
- Article: not found
Fusobacterium nucleatum Promotes Chemoresistance to Colorectal Cancer by Modulating Autophagy
- Record: found
- Abstract: found
- Article: not found
The sympathetic control of blood pressure.
- Record: found
- Abstract: found
- Article: not found